Join us on Thursday September 24th at 2 p.m. for a fun filled session of Advanced STEM Club where we will learn even more about physical and chemical changes and get to do a fun experiment together! WARNING this experiment should ONLY be done under adult supervision using proper safety gear and in a very well ventilated area, preferably outdoors.
Many, but not all, metals react with acids. Hydrogen gas forms as the metals react with the acid to form salts. This means that you can use acids to dissolve most metals, this depends on the kind of metal used and the concentration, or strength, of the acid used. To understand how this works we must first understand what a solution is and what we mean by dissolving.
The following information comes from Ducksters:
What is a solution?
A solution is a specific type of mixture where one substance is dissolved into another. A solution is the same, or uniform, throughout which makes it a homogeneous mixture . Go here to learn more about mixtures.
A solution has certain characteristics:
- It is uniform, or homogeneous, throughout the mixture
- It is stable and doesn’t change over time or settle
- The solute particles are so small they cannot be separated by filtering
- The solute and solvent molecules cannot be distinguished by the naked eye
- It does not scatter a beam of light
Example of a Solution
One example of a solution is salt water which is a mixture of water and salt. You cannot see the salt and the salt and water will stay a solution if left alone.
Parts of a Solution
- Solute – The solute is the substance that is being dissolved by another substance. In the example above, the salt is the solute.
- Solvent – The solvent is the substance that dissolves the other substance. In the example above, the water is the solvent.
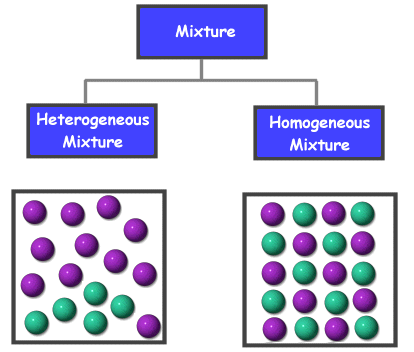
A solution is a type of homogeneous mixture
Dissolving
A solution is made when one substance called the solute “dissolves” into another substance called the solvent. Dissolving is when the solute breaks up from a larger crystal of molecules into much smaller groups or individual molecules. This break up is caused by coming into contact with the solvent.
In the case of salt water, the water molecules break off salt molecules from the larger crystal lattice. They do this by pulling away the ions and then surrounding the salt molecules. Each salt molecule still exists. It is just now surrounded by water molecules instead of fixed to a crystal of salt.
Now that we know about solutions we need to understand physical and chemical changes. The following information is from ThoughtCo:
In a nutshell, a chemical change produces a new substance, while a physical change does not. A material may change shapes or forms while undergoing a physical change, but no chemical reactions occur and no new compounds are produced.
Key Takeaways: Chemical and Physical Change Examples
- A chemical change results from a chemical reaction, while a physical change is when matter changes forms but not chemical identity.
- Examples of chemical changes are burning, cooking, rusting, and rotting.
- Examples of physical changes are boiling, melting, freezing, and shredding.
- Often, physical changes can be undone, if energy is input. The only way to reverse a chemical change is via another chemical reaction.
Examples of Chemical Changes
A new compound (product) results from a chemical change as the atoms rearrange themselves to form new chemical bonds.
- Burning wood
- Souring milk
- Mixing acid and base
- Digesting food
- Cooking an egg
- Heating sugar to form caramel
- Baking a cake
- Rusting of iron
Examples of Physical Changes
No new chemical species forms in a physical change. Changing the state of a pure substance between solid, liquid, and gas phases of matter are all physical changes since the identity of the matter does not change.
- Crumpling a sheet of aluminum foil
- Melting an ice cube
- Casting silver in a mold
- Breaking a bottle
- Boiling water
- Evaporating alcohol
- Shredding paper
- Sublimation of dry ice into carbon dioxide vapor
How to Tell Whether It’s a Physical or Chemical Change?
Look for an indication that a chemical change occurred. Chemical reactions release or absorb heat or other energy or may produce a gas, odor, color or sound. If you don’t see any of these indications, a physical change likely occurred. Be aware a physical change may produce a dramatic change in the appearance of a substance. This doesn’t mean a chemical reaction occurred.
So is Dissolving Metal a Chemical or Physical Change?
The following information comes from Sciencing: Dissolving metals is a chemical property that takes place when water or strong acids react with metallic objects. Chemical forces pull metal atoms from the object, causing it to break apart and leave the atoms floating freely in solution. Solubility depends on the acids and metals involved. Lead and iron react easily, while platinum and gold are much harder to dissolve.
Why Metals Dissolve
Metals possess a chemical property called “activity” because of their ability to lose electrons to other atoms. An activity series ranks metals by how reactive they are, with sodium and lithium ranking very high and gold having a low rank. In water or acids, the metals trade places with hydrogen. The hydrogen escapes as a gas, and the metal atoms, no longer attached to the object from which they came, dissolve in solution.
Acids
Strong acids are a combination of hydrogen and an element or compound called a conjugate base. For example, hydrochloric acid pairs hydrogen and chlorine as HCl. When the acid dissolves in water, hydrogen splits off from the base and the solution becomes a powerful solvent. Hydrochloric acid dissolves the less active metals, such as zinc and magnesium, easily. It dissolves the more-resistant iron, copper and related metals less easily, or not at all. Other chemicals, such as nitric acid, will dissolve some metals that hydrochloric acid will not.
Water
The most active metals, which include sodium and potassium, dissolve instantly and dramatically in plain water — no stronger acid is needed. The metals react violently with water, releasing and igniting hydrogen gas and causing an explosion. Because of the strong reactivity of these metals with water, it is dangerous to leave them exposed even to the moisture in humid air. They are usually stored in mineral oil, with which they do not react.
Noble Metals
A class of elements called the noble or precious metals dissolve only with difficulty. Platinum, iridium, gold and osmium, in particular, stand up to attack by strong hydrochloric and nitric acids. However, by carefully combining them, you obtain a powerful solvent known as aqua regia, which does dissolve gold. Platinum and a few other metals are particularly resistant to acids, but hot aqua regia dissolves them, although slowly.
Now that we understand the science behind our experiment today it’s time to dissolve some metal!
Materials:
Procedure:
- Make sure that you are in a well ventilated area, preferably outdoors, as this experiment will release a small amount of gasses that can be toxic (and smell real bad).
- Put on your safety glasses and gloves.
- Very carefully pour the drain cleaner into your bowl. Try to avoid splashing while pouring by going slowly. Fill the bowl until there is about an inch of liquid.
- Using your tongs place a square (around 2×2 inches) into the bowl. Depending on how strong your drain cleaner is you will start to see a reaction anywhere from immediately to within 1 minute. Depending on your drain cleaner your aluminum foil will completely dissolve within 10 minutes, the stronger the drain cleaner the faster the foil will dissolve.
- If you want to you can try placing other metals in the drain cleaner such as a penny, a paper clip, or the tab from a soda can.
Additional Resources:
- PBS Learning Media
- PBS Learning Media 2
- Royal Society of Chemistry
- Sciencing – Dissolving Steel
- Sciencing – Dissolving Metals
- Doctor Mad Science
- Ducksters
- State Government of Victoria, Australia – Science and STEM Teaching Materials and Methods
- American Chemical Society
- ThoughtCo
- LibreTexts – This is a great resource for free open source textbooks for older students.
- Chem4Kids










